Join us now! Explore what it is like to work in wOmics!











WESTLAKE OMICS

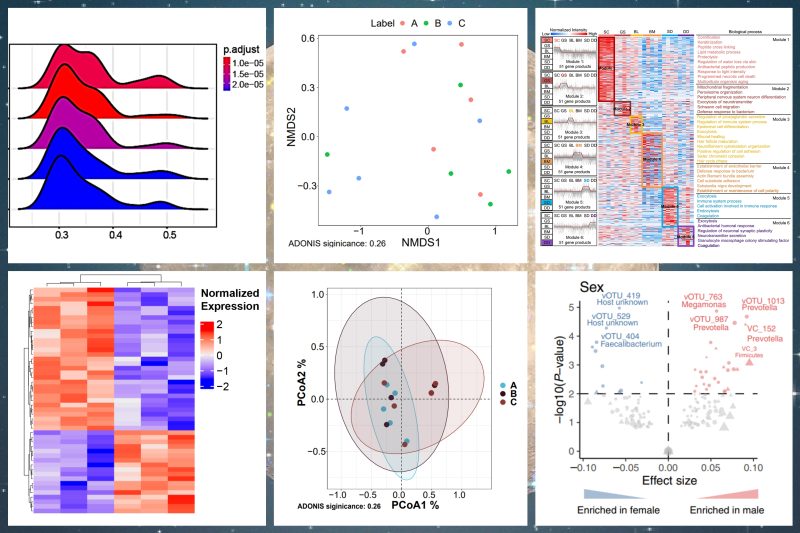
Established in July 2020, Westlake Omics is a micro-world data company empowered by Al. Its primary goal is to enhance precision medicine and facilitate drug research and development by leveraging technological innovations in proteomics and multimodal big data technology.
Westlake Omics has partnered with more than 100 prestigious universities, research institutes, hospitals, and pharmaceutical companies both domestically and internationally. Additionally, it holds ownership of multiple national patents and software copyrights.














-5x3.jpeg)

